Urgent warning: Positive PCR detection results for infectious myonecrosis virus (IMNV) and decapod iridescent virus 1 (DIV1) in captured Penaeus monodon from the Indian Ocean
25 February 2020 | Jiraporn Srisala, Piyachat Sanguanrut, Dararat Thaiue, Saensook Laiphrom, Jittima Siriwattano, Juthatip Khudet, Sorawit Powtongsook, Timothy W. Flegel, Kallaya Sritunyalucksana | 1202 Downloads | .pdf | 81.59 KB | Health and welfare, Shrimp
NACA would like to thank the authors for sharing this information with the network - Ed.
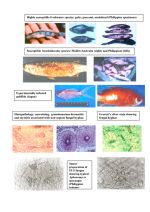
From a survey of wild, adult Penaeus monodon of potential broodstock size from the Indian Ocean in April 2018, we obtained positive nested RT-PCR test results for infectious myonecrosis virus (IMNV) (2/26 shrimp in one specimen lot) and positive nested PCR test results for DIV1 (5/26 shrimp in a different lot). The test results were obtained using nucleic acid extracted from pleopods (swimming legs) and the PCR protocols used were those previously published for SHIV (Qiu, et al., 2017) and IMNV (Poulos Lightner, 2006; Poulos, et al., 2006); (Senapin, et al., 2007). The amplicon sequences from these tests were 99-100% identical to the matching regions published for the two viruses. As a confirmatory step, a second round of nested PCR tests was carried out using new, in-house primers designed from regions of the respective viral genomes distant from the target regions used in the first round of tests. These new tests had never been used previously in our laboratory and the positive and negative results for both viruses corresponded with those for the same individual specimens from the first round of testing for the partner target gene. Again, the amplicon sequences were 99-100% identical to the matching regions published for the two viruses.
These results suggested the possibility that the grossly normal, PCR-positive captured P. monodon specimens might be infected with the respective viruses at the carrier level. If so, they might serve as potential vehicles for introduction of IMNV and/or DIV1 into crustacean culture systems, especially if they were used in hatcheries for production of PL for distribution to shrimp farmers without proper precautions in place.
We recommend that wild, captured P. monodon from the Indian Ocean intended for use as broodstock be subjected to PCR testing before use in a hatchery and that they be discarded, if they are found to be positive. If not positive, their larvae and post-larvae (PL) should be monitored for presence of these 2 viruses periodically during production and again before they are sold to users. We also strongly recommend that industry practitioners using wild, captured P. monodon be discouraged from handling it together with broodstock of other crustaceans listed above in common maturation or hatchery facilities. We also recommend that shrimp farmers be discouraged from cultivating those species together with P. monodon in the same pond or on the same farm, especially if the latter originated from wild, captured broodstock that have not been tested for freedom from IMNV and/or DIV1 as applicable based on susceptibility of the specific species. Indeed, since domesticated stocks of P. monodon SPF for IMNV and DIV1 are available, we do not recommend the use of captured wild P. monodon broodstock for PL production at all.
Publisher: Network of Aquaculture Centres in Asia-Pacific
Rights: Copyright, all rights reserved.